Turbine isentropic efficiency
Turbine isentropic efficiency#
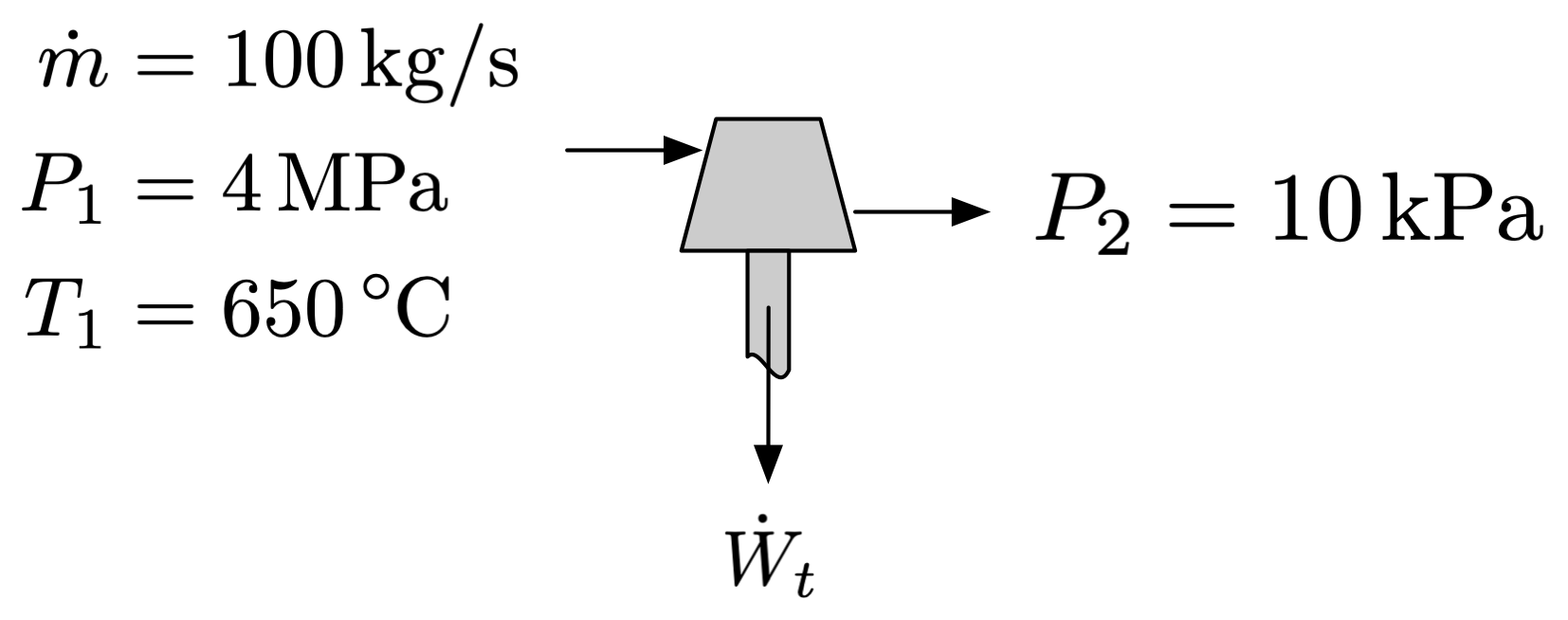
A steam turbine performs with an isentropic efficiency of \(\eta_t = 0.84\). The inlet conditions are 4 MPa and 650°C, with a mass flow rate of 100 kg/s, and the exit pressure is 10 kPa. Assume the turbine is adiabatic.
Problem:
Determine the power produced by the turbine
Determine the rate of entropy generation
import numpy as np
import cantera as ct
from pint import UnitRegistry
ureg = UnitRegistry()
Q_ = ureg.Quantity
We can start by specifying state 1 and the other known quantities:
temp1 = Q_(650, 'degC')
pres1 = Q_(4, 'MPa')
state1 = ct.Water()
state1.TP = temp1.to('K').magnitude, pres1.to('Pa').magnitude
mass_flow_rate = Q_(100, 'kg/s')
efficiency = 0.84
pres2 = Q_(10, 'kPa')
To apply the isentropic efficiency, we’ll need to separately consider the real turbine and an equivalent turbine operating in a reversible manner. They have the same initial conditions and mass flow rate.
For the reversible turbine, an entropy balance gives:
and then with \(P_2\) and \(s_{s,2}\) we can fix state 2 for the reversible turbine:
state2_rev = ct.Water()
state2_rev.SP = state1.s, pres2.to('Pa').magnitude
state2_rev()
water:
temperature 319 K
pressure 10000 Pa
density 0.07464 kg/m^3
mean mol. weight 18.016 kg/kmol
vapor fraction 0.91292
phase of matter liquid-gas-mix
1 kg 1 kmol
--------------- ---------------
enthalpy -1.3594e+07 -2.4492e+08 J
internal energy -1.3728e+07 -2.4733e+08 J
entropy 11017 1.9849e+05 J/K
Gibbs function -1.7109e+07 -3.0824e+08 J
heat capacity c_p inf inf J/K
heat capacity c_v nan nan J/K
Then, we can do an energy balance for the reversible turbine, which is also steady state and adiabatic:
Then, recall that the isentropic efficiency is defined as
so we can obtain the actual turbine work using \(\dot{W}_t = \eta_t \dot{W}_{s,t}\) :
work_isentropic = mass_flow_rate * (
Q_(state1.h, 'J/kg') - Q_(state2_rev.h, 'J/kg')
)
work_actual = efficiency * work_isentropic
print(f'Actual turbine work: {work_actual.to(ureg.megawatt): .2f}')
Actual turbine work: 118.73 megawatt
Then, we can perform an energy balance on the actual turbine:
which we can use with the exit pressure to fix state 2.
enthalpy2 = Q_(state1.h, 'J/kg') - (work_actual / mass_flow_rate)
state2 = ct.Water()
state2.HP = enthalpy2.to('J/kg').magnitude, pres2.to('Pa').magnitude
state2()
water:
temperature 328.39 K
pressure 10000 Pa
density 0.066165 kg/m^3
mean mol. weight 18.016 kg/kmol
vapor fraction 1
phase of matter gas
1 kg 1 kmol
--------------- ---------------
enthalpy -1.3368e+07 -2.4084e+08 J
internal energy -1.3519e+07 -2.4357e+08 J
entropy 11725 2.1124e+05 J/K
Gibbs function -1.7219e+07 -3.1021e+08 J
heat capacity c_p 1894.4 34129 J/K
heat capacity c_v 1423 25637 J/K
Finally, we can perform an entropy balance on the actual turbine:
which allows us to find the rate of entropy generation.
entropy_gen = mass_flow_rate * (
Q_(state2.s, 'J/(kg K)') - Q_(state1.s, 'J/(kg K)')
)
print(f'rate of entropy generation: {entropy_gen.to("kW/K"): .2f}')
rate of entropy generation: 70.81 kilowatt / kelvin