Reduced Helmholtz equation of state: carbon dioxide
Contents
Reduced Helmholtz equation of state: carbon dioxide#
Water equation of state: You can see the full, state-of-the-art equation of state for water, which also uses a reduced Helmholtz approach: the IAPWS 1995 formulation [WPruss02]. This equation is state is available using CoolProp with the Water fluid.
One modern approach for calculating thermodynamic properties of real fluids uses a reduced Helmholtz equation of state, using the reduced Helmholtz free energy function \(\alpha\):
which is a function of reduced density \(\delta\) and reduced temperature \(\tau\):
The reduced Helmhotz free energy function, \(\alpha(\tau, \delta)\), is given as the sum of ideal gas and residual components:
which are both given as high-order fits using many coefficients:
import matplotlib.pyplot as plt
%matplotlib inline
# these are mostly for making the saved figures nicer
import matplotlib_inline.backend_inline
matplotlib_inline.backend_inline.set_matplotlib_formats('pdf', 'png')
plt.rcParams['figure.dpi']= 300
plt.rcParams['savefig.dpi'] = 300
import numpy as np
import cantera as ct
from scipy import integrate, optimize
from pint import UnitRegistry
ureg = UnitRegistry()
Q_ = ureg.Quantity
import sympy
sympy.init_printing(use_latex='mathjax')
T, R, tau, delta = sympy.symbols('T, R, tau, delta', real=True)
a_vars = sympy.symbols('a0, a1, a2, a3, a4, a5, a6, a7', real=True)
theta_vars = sympy.symbols('theta3, theta4, theta5, theta6, theta7', real=True)
n_vars = sympy.symbols('n0, n1, n2, n3, n4, n5, n6, n7, n8, n9, n10, n11', real=True)
alpha_ideal = sympy.log(delta) + a_vars[0] + a_vars[1]*tau + a_vars[2]*sympy.log(tau)
for i in range(3, 8):
alpha_ideal += a_vars[i] * sympy.log(1.0 - sympy.exp(-tau * theta_vars[i-3]))
display(sympy.Eq(sympy.symbols('alpha_IG'), alpha_ideal))
alpha_res = (
n_vars[0] * delta * tau**0.25 +
n_vars[1] * delta * tau**1.25 +
n_vars[2] * delta * tau**1.50 +
n_vars[3] * delta**3 * tau**0.25 +
n_vars[4] * delta**7 * tau**0.875 +
n_vars[5] * delta * tau**2.375 * sympy.exp(-delta) +
n_vars[6] * delta**2 * tau**2 * sympy.exp(-delta) +
n_vars[7] * delta**5 * tau**2.125 * sympy.exp(-delta) +
n_vars[8] * delta * tau**3.5 * sympy.exp(-delta**2) +
n_vars[9] * delta * tau**6.5 * sympy.exp(-delta**2) +
n_vars[10] * delta**4 * tau**4.75 * sympy.exp(-delta**2) +
n_vars[11] * delta**2 * tau**12.5 * sympy.exp(-delta**3)
)
display(sympy.Eq(sympy.symbols('alpha_res'), alpha_res))
Carbon dioxide equation of state#
The coefficients \(a_i\), \(\theta_i\), and \(n_i\) are given for carbon dioxide:
# actual coefficients
coeffs_a = [
8.37304456, -3.70454304, 2.500000, 1.99427042,
0.62105248, 0.41195293, 1.04028922, 8.327678e-2
]
coeffs_theta = [
3.151630, 6.111900, 6.777080, 11.32384, 27.08792
]
coeffs_n = [
0.89875108, -0.21281985e1, -0.68190320e-1, 0.76355306e-1,
0.22053253e-3, 0.41541823, 0.71335657, 0.30354234e-3,
-0.36643143, -0.14407781e-2, -0.89166707e-1, -0.23699887e-1
]
Through some math, we can find an expression for pressure:
Use this expression to estimate the pressure at \(T\) = 350 K and \(v\) = 0.01 m\(^3\)/kg, and compare against that obtained from Cantera. We can use our symbolic expression for \(\alpha_{\text{res}} (\tau, \delta)\) and take the partial derivative:
# use Cantera fluid to get specific gas constant and critical properties
f = ct.CarbonDioxide()
gas_constant = ct.gas_constant / f.mean_molecular_weight
temp_crit = f.critical_temperature
density_crit = f.critical_density
# conditions of interest
temp = 350
specific_volume = 0.01
density = 1.0 / specific_volume
# take the partial derivative of alpha_res with respect to delta
derivative_alpha_delta = sympy.diff(alpha_res, delta)
# substitute all coefficients
derivative_alpha_delta = derivative_alpha_delta.subs(
[(n, n_val) for n, n_val in zip(n_vars, coeffs_n)]
)
def get_pressure(
temp, specific_vol, fluid, derivative_alpha_delta, tau, delta
):
'''Calculates pressure for reduced Helmholtz equation of state'''
red_density = (1.0 / specific_vol) / fluid.critical_density
red_temp_inv = fluid.critical_temperature / temp
gas_constant = ct.gas_constant / fluid.mean_molecular_weight
dalpha_ddelta = derivative_alpha_delta.subs(
[(delta, red_density), (tau, red_temp_inv)]
)
pres = (
gas_constant * temp * (1.0 / specific_vol) *
(1.0 + red_density * dalpha_ddelta)
)
return pres
pres = get_pressure(temp, specific_volume, f, derivative_alpha_delta, tau, delta)
print(f'Pressure: {pres / 1e6: .3f} MPa')
f.TV = temp, specific_volume
print(f'Cantera pressure: {f.P / 1e6: .3f} MPa')
Pressure: 5.464 MPa
Cantera pressure: 5.475 MPa
Our calculation and that from Cantera agree fairly well! They are not exactly the same because Cantera uses a slightly different formulation for the equation of state.
Let’s compare the calculations now for a range of specific volumes and multiple temperatures:
fig, ax = plt.subplots(figsize=(8, 4))
specific_volumes = np.geomspace(0.001, 0.01, num=20)
temperatures = [300, 400, 500]
for temp in temperatures:
pressures = np.zeros(len(specific_volumes))
pressures_cantera = np.zeros(len(specific_volumes))
for idx, spec_vol in enumerate(specific_volumes):
pressures[idx] = get_pressure(
temp, spec_vol, f,
derivative_alpha_delta, tau, delta
)
f.TV = temp, spec_vol
pressures_cantera[idx] = f.P
ax.loglog(specific_volumes, pressures/1000., 'o', color='blue')
ax.loglog(specific_volumes, pressures_cantera/1000., color='blue')
bbox_props = dict(boxstyle='round', fc='w', ec='0.3', alpha=0.9)
ax.text(2e-3, 4e3, '300 K', ha='center', va='center', bbox=bbox_props)
ax.text(2e-3, 1.6e4, '400 K', ha='center', va='center', bbox=bbox_props)
ax.text(2e-3, 7e4, '500 K', ha='center', va='center', bbox=bbox_props)
ax.legend(['Reduced Helmholtz', 'Cantera'])
plt.grid(True, which='both')
plt.xlabel('Specific volume (m^3/kg)')
plt.ylabel('Pressure (kPa)')
fig.tight_layout()
plt.show()
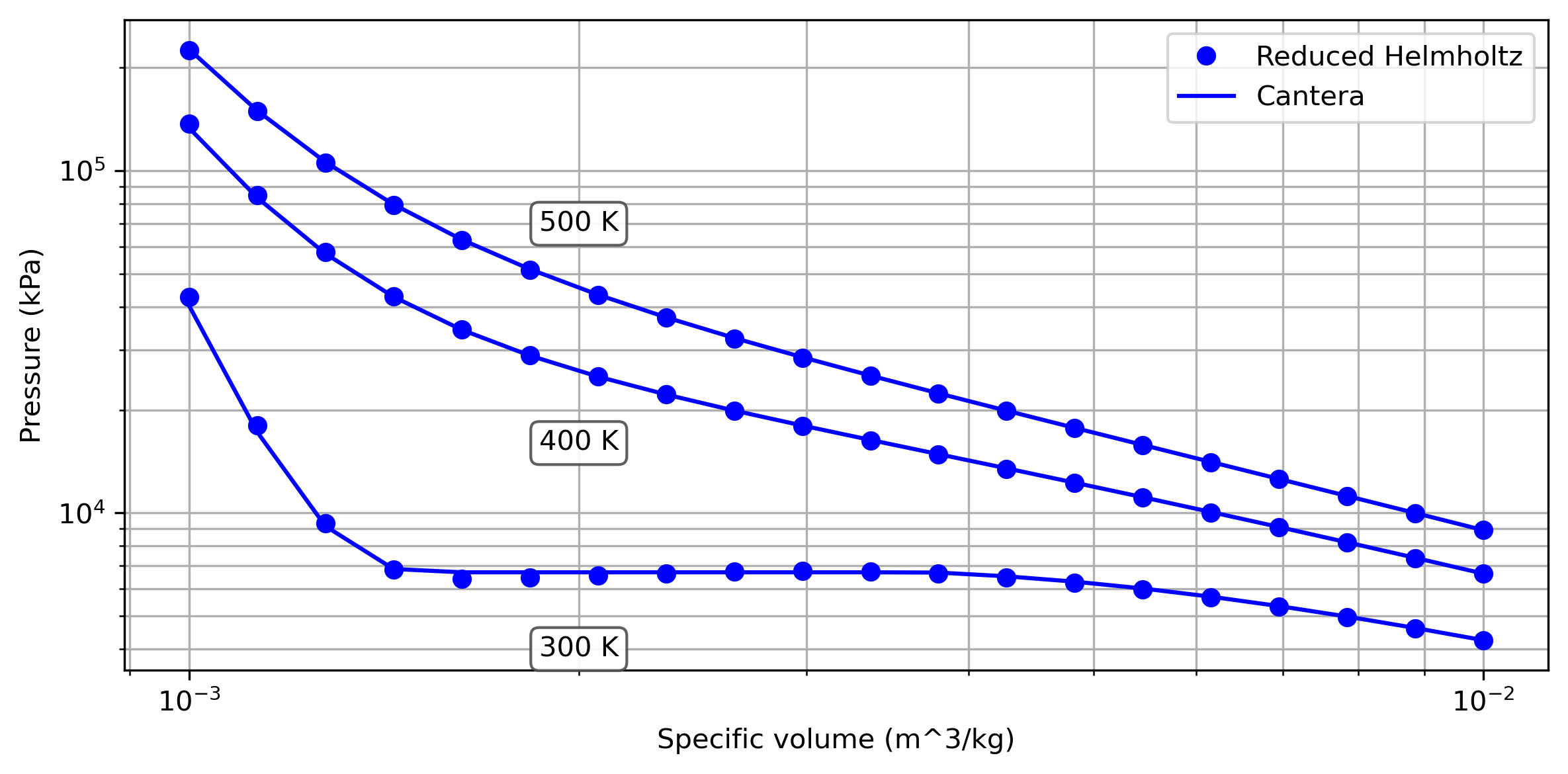
We can see that the pressure calculated using the reduced Helmholtz equation of state matches closely with that from Cantera, which uses a different but similarly advanced equation of state.